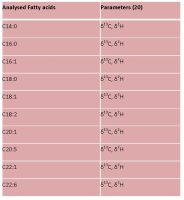
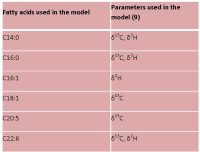
Pesticides and other chemical compounds are widely used in agriculture because they protect crops and improve the efficiency of food production. However, it is necessary to identify what type and how much chemical residues are in food, water and soil as these residues may pose a potential threat to human health as well as the environment. Reducing pesticides in food will result in a lower toxic chemical burden entering the body and accumulating in the tissues and organs, but it will take a concerted effort.
The European Union (EU) member states are implementing extensive policy changes to improve soil quality and ultimately improve the quality of crops. Among them is a “proposal for a new Regulation on the Sustainable Use of Plant Protection Products, including EU wide targets to reduce by 50% the use and risk of chemical pesticides by 2030.” In July 2023, the European Commission also “adopted a package of measures for a sustainable use of key natural resources, which will also strengthen the resilience of EU food systems and farming.”
Reducing the use of chemicals is an important step to ensuring enough safe food for the growing human population. This challenge comes at a time when arable land is being lost, the demand for food is increasing and the world population is expected to increase to 9.7 billion in 2050, according to the United Nations. This will require food growers and processors to implement more sustainable growing practices often referred to as Integrated Pest Management (IPM). While the benefits of natural pest control are already well understood—clean water, healthy soil and improved biodiversity—reducing reliance on synthetic pesticides will require increased analysis, and as a result, generating a lot of data.
It all begins with the analysis of chemical residues found on crops.
Naming the Culprits
With more than 1,000 pesticides in use around the world, it is important to know the properties and toxicological effects of each. A group of pesticides commonly used to curb weeds is herbicides. Glyphosate, (N-(phosphonomethyl)glycine), a widely-used, broad-spectrum, systemic herbicide and crop desiccant, has in the recent years come under scrutiny as the International Agency for Research on Cancer (IARC), a branch of the World Health Organization, classified glyphosate as “probably carcinogenic to humans.”
In the coming years, more data will be gathered on pesticides in the EU, with part of that information coming from control measures and agricultural practice reviews. A large part will come from laboratory measurements to meet data requirements mandated by the new regulations. Compiling consistent, accurate data depends on the equipment that produces it. This is particularly important for food safety.
As technologies advance and more information can be obtained, including residues on food, the requirements for the type of data are also changing. For example, quantifying how much of a predetermined pesticide residue is in a sample is a narrow parameter. Identifying all of the compounds that can be found will provide more data to characterize the sample. Ideally, collecting both will provide the most complete answer to the question, “How much and what kinds of pesticides remain on our food?”
Once that information is available, it is possible to choose appropriate remediation steps. But it takes sensitive laboratory equipment to both identify and quantify residues.
Connecting the Dots Between Pesticides and Food
The most common technology currently used to monitor pesticides is liquid chromatography-mass spectrometry (LC-MS). First, the sample (e.g., soil, water, fruit or vegetable) is injected onto the liquid chromatograph (LC). The LC separates the complex mixture based on the chemical properties of the individual pesticides before being analyzed by the mass spectrometer (MS). MS instruments analyze samples based their masses—or more correctly their mass-to-charge ratio—in a very accurate and precise manner. MS/MS instruments also break apart the pesticides and are able to look for these fragments. These are used to quickly determine if a specific compound is present and in what amount, known as identification and quantitation.
Amadeo Rodríguez Fernández-Alba, professor in analytical chemistry at the University of Almeria and head of the European Reference Laboratory for Pesticide Residues, has valued the benefits of using LC-MS/MS for pesticide residue analysis for years. Over time, the technology and methods have evolved to identify and measure the amount of chemicals in food plants and soil.
In his recent work, Fernández-Alba showed the analysis of 30 compounds of emerging concern (CECs) in soils irrigated with simulated reclaimed water on trial farmland using a targeted MS/MS approach. An accumulation of 13 pesticides and 5 pharmaceuticals could be found at different rates, highlighting the importance of increased analysis for reclaimed water testing.
Regarding the testing method, the authors pointed out that, “a modified QuEChERS method showed the best results in terms of extractability and accuracy. The extraction procedure developed provided adequate extraction performances (70% of the target analytes were recovered within a 70–99% range), with good repeatability and reproducibility (variations below 20%) and great sensitivity (LOQ < 0.1 ng/g in most cases). No matrix effects were observed for 70% of the compounds. Finally, the analytical methodology was applied in a pilot study where agricultural soil was irrigated with reclaimed water spiked with the contaminants under study. Of the 25 CECs added in irrigation water, a total of 13 pesticides and 5 pharmaceutical products were detected…”
Reduction in pesticide usage needs to be monitored in both field and food samples for a wide range of analytes including unknown substances to build confidence in the food system. Using mass spectrometry can provide that data.
Glyphosate, mentioned above, is one of the most widely used agrochemicals in the world and also one of the most difficult to detect. In Europe, EFSA has proposed MRLs for a wide range of commodities for glyphosate. Monitoring this kind of highly polar, small-organic pesticide in food and water from diverse sources can be complex, time-consuming and expensive. NofaLab, a sampling and testing lab in the Netherlands, collaborated with SCIEX to create a high throughput method using LC-MS/MS to test for as many polar pesticides in a single analysis as possible.
The final method utilized the sensitivity of QTRAP technology and was “found to be considerably more robust and sensitive than other approaches described in various publications and have achieved the target limits of detection required to meet existing and proposed future regulations.” In addition, the “ion chromatographic approach to the analysis of polar pesticides offers the ability to include multiple analytes in a single injection without derivatization…allowing high-throughput laboratories to manage samples efficiently and minimize running costs.”
Targeted MS/MS analysis has long been the gold standard for pesticide analysis in the industry, but advanced high-resolution MS systems enable even greater accuracy and confidence, helping to identify more contaminants, even unknowns. The ZenoTOF 7600 system uses electron activated dissociation (EAD) to create a higher number of fragments as compared to collision induced fragmentation (CID), which is traditionally used in targeted MS/MS analysis on other systems, allowing for highly confident identifications of pesticides in food samples. The ZenoTrap technology additionally enhances sensitivity, which is needed in pesticide analysis to meet regulatory limits. Regardless of the type of sample, whether taken from the soil in a field or from harvested crops, mass spectrometers can identify the type and amount of chemicals present in a sample within several minutes of run time and analyze hundreds of samples in a day.
Streamlining Data Review and Adhering to Data Standards
In an effort to standardize pesticide use, the Codex Alimentarius Commission (CAC) established standards for pesticide residues and developed international standards for food products. This framework for providers at various points in the food supply chain can help reduce the risk of contamination and toxicity.
In the U.S., the Environmental Protection Agency (EPA) establishes tolerances, also known as maximum residue limits (MRL) in other countries, for the type and quantity of pesticides that can remain on food. The agency sets these to ensure pesticides can be used with “reasonable certainty of no harm.”
Producers adhering to these guidelines must handle and present extensive data sets from test results, and any new future regulations will require robust data to track the success of the initiatives and effectively enforce their use. Reviewing and understanding data in order to make decisions is tricky and labor-intensive. It can take laboratories hours every day to process, interpret and manage the data. Software enables fast data processing and fast review by exception flagging, which is valuable in food safety laboratories that typically see a high turn-around in samples every day.
Maintaining resilient food systems will be rooted in data-driven decisions that improve food safety, including limiting pesticide and other chemical uses. By using modern mass spectrometry technology, researchers can be more confident that their food analyses will lead to better-informed policies, more sustainable agricultural practices, and healthier food for future generations.