
Public-Private Partnership Aims to Fill Gaps in Supply Chain Interoperability
Partnership for Food Traceability seeking members to play key roles in decision-making.
Partnership for Food Traceability seeking members to play key roles in decision-making.

Maintaining the safety of the food we consume is complex and multi-faceted. Inspections and audits have become the backbone of our food industry’s quality control processes in preventing foodborne illnesses and ensuring the stringent regulatory measures are met. This article details the intricacies of the importance of inspections and audits, highlighting the procedure involved to reduce the consistency of recalls.

The researchers reported that 43, 35, and 0% of the products tested exceed Prop 65 MADLs for heavy metal concentrations.
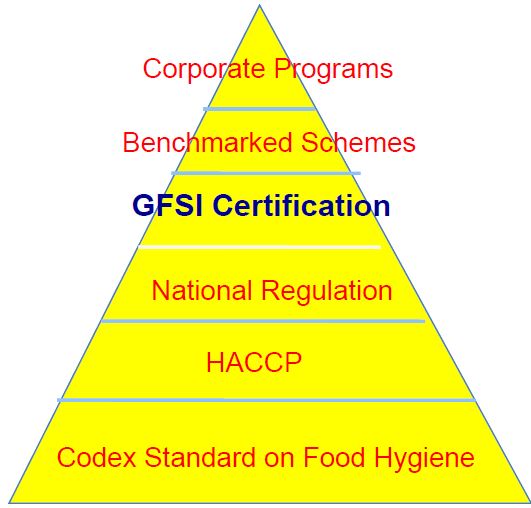
This paper compares U.S. food laws with GFSI standards, highlighting their respective strengths and weaknesses. U.S. food laws provide a legally enforceable, standardized framework that ensures consumer protection and public accountability. Conversely, GFSI standards offer flexibility, global recognition, and industry-driven innovation, albeit at higher costs and with less enforcement power. The U.S. food safety system, despite its complexity, delivers robust protection through comprehensive federal regulations. A balanced approach that integrates the strengths of both systems could optimize food safety, leveraging legal enforcement and global industry standards.

A gap assessment can help determine what requirements existing traceability programs already meet and identify where improvements are needed to comply with the final Food Traceability Rule by the January 2026 deadline.

FDA is seeking feedback. There is only one week left to submit your comments to the docket on the New Era of Smarter Food Safety.

The GFSI Steering Committee has suspended recognition of certification programmes owned by the Equitable Food Initiaive until evidence of re-alignment to GFSI Benchmarking requirements v 2020 is provided by the Certification Programme Owner EFI. This suspension took effect on 7th June 2024.
With the effective date for updated traceability recordkeeping approaching in January 2026, traceability is a top priority for most organizations working in the food industry. Produce companies are especially impacted by traceability requirements as the first step in the food supply chain.

Food allergens present a real risk to modern consumers. As a producer, you can clean up your act by abiding by FSMA guidelines, implementing automation, and improving your employee training.

The USDA, FDA and HHS have announced efforts to combat the spread of H5N1 and provide financial support for lost milk production.